SOP
Management System
 Pharmaceutical Pharmaceutical |
 Laboratory Laboratory |
 Healthcare Healthcare |
| GMP Compliance | CAP Compliance | JCI Compliance |
| FDA Compliance | NABL Compliance | NABH Compliance |
Standard Operating Procedure (SOP) is at the heart of compliance for Pharmaceutical, Laboratory and Healthcare organizations.
CCE Software understands that though similar, the three industries have different compliance regulations. CCE Software therefore offers three distinct SOP Management Systems, covering the entire life-cycle of SOP.
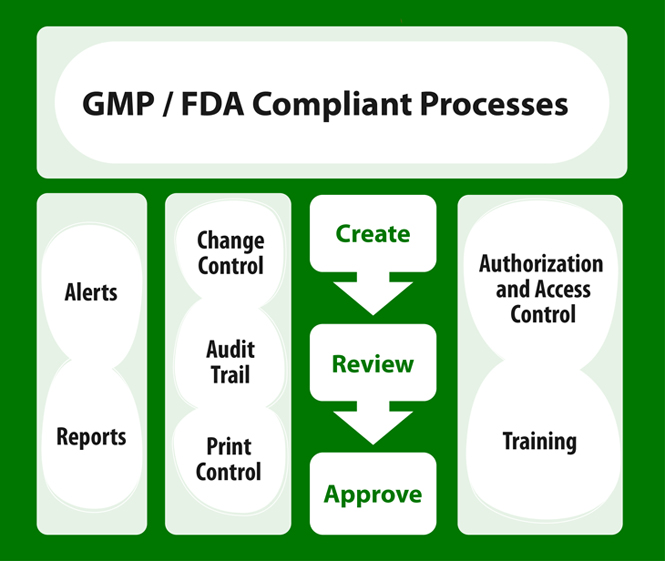
GMP/FDA Compliant SOP Management for Pharmaceuticals
SOP software for pharmaceuticals. Specifically designed SOP Software for GMP and US FDA compliance.
SOP software for Laboratory. Created as per the requirement of SOP Software for CAP and NABL compliance of life-science / pathological and other test labs.
SOP software for hospitals and healthcare units. The organizations engaged in healthcare, be it hospitals, clinics, etc. require SOP Software with JCI or NABH compliance.
CCE Software’s SOP Management System has the 3 variants catering to the focused need of the 3 types of organizations.
For each of the 3 variants, SOP Management System has unique built-in processes.
ADVANTAGES:
SOP Creation as Per Regulatory Compliant Process
- Creation of SOPs with Automatic Notifications and Seamless Review Workflows
Easy Management of SOP Life-cycle
- Complete Control of SOPs by QA
Easy Change Control and Version Control as Per Regulation
- Compliant Change Control, Authorized Tracking of The History of SOP Versions.
Audit Made Easy
- Easy to Face Regulatory Audit
Print Control
- Total Control over Printing SOP Documents to Avoid any Non-Compliance
Reports and Timely Alerts
- Timely alerts helps compliance. Wide Range of Reports at The Click of a Button
